New microscope images single, living cells at better resolution and lower light dose
Computed tomography (CT) is benefiting from research to lower radiation dose, while maintaining or improving the quality of the images. Analogously, scientists at the National Institute of Biomedical Imaging and Bioengineering (NIBIB), part of the National Institutes of Health (NIH), and the University of Chicago have developed a new microscope that doubles the resolution of images without exposing the sample to an increased amount of light or prolonging the imaging process.
Fluorescence imaging is generally inefficient, as the majority of the light emitted from the biological sample does not get recorded. This means that researchers need to collect more light over a longer time to improve the clarity of the picture. It is not unlike the problem photographers face when they try to take a photo in low light. Usually they have to choose between increasing the amount of light drastically by using a flash — or keeping the shutter open for longer than usual. However, sensitive biological samples, such as individual cells or worm embryos, are highly sensitive to light and can be damaged or even killed by traditional microscopy, which uses strong light for an extended period of time.
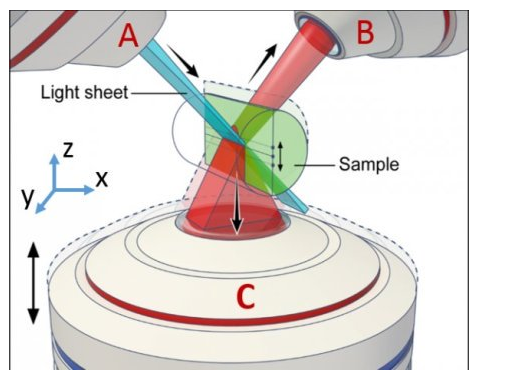
Previously, Yicong Wu, Ph.D., Staff Scientist, and Hari Shroff, Ph.D., Chief of the NIBIB High Resolution Optical Imaging team developed new microscopes to reduce the amount of light needed to image biological sample. Now, they’ve added another lens to supplement their earlier dual-view microscope. The new lens images the sample from below, thus capturing even more light emitted from the sample.
Wu and Shroff then collaborated with Patrick La Riviere, Ph.D., of the University of Chicago’s Radiology Department. Riviere’s research focuses on producing algorithms that allow radiologists to use less ionizing radiation in CT scans on humans. Together with Riviere’s team, they were able to design and implement algorithms that merge the three images into one (a process called deconvolution) — creating a sharper, clearer 3D image than previously possible.
“Our lab is composed of physicists, engineers, and biologists. In this case, collaboration with Patrick, an expert on computational imaging, was essential in developing the analysis that properly merged the multiple views of the microscope into one,” says Shroff. “The tools we built can be extended to other microscopes, and we look forward to continuing our productive collaboration. Cross-disciplinary collaborative research takes more time, but has the potential to uncover biological insights missed by more traditional approaches.”
The microscope doubles the resolution of fluorescence microscopy. As medical researchers become ever more interested in how individual cells work, studying living organisms requires technology that can help scientists actually see how some of the smallest functions in the human body work. For example, being able to see how a virus enters a cell, and how it moves around once inside, could go a long way towards scientists’ understanding of how infections occur and potentially how to fight them more effectively.
“Having better tools is important no matter what you’re doing, from carpentry to scientific research,” said Shroff. “The microscopes being developed here, along with concurrent advances in computational imaging, help to give researchers the tools they need to solve some of medicine’s most challenging problems.”
The work is published in the August 11 issue of Optica.
https://www.sciencedaily.com/releases/2016/09/160901130029.htm